
Dr. Vivien Brown
Family Physician, Assistant Professor, Faculty of Medicine, University of Toronto
Dr. Beverly Francis
PhD, Director of Scientific Leadership — North America at Moderna
In the fight against COVID-19, Moderna stays ahead of the science with its mRNA platform, while also developing vaccine technology in other therapeutic areas.
The fall season is upon us and with it brings a lot of unknowns about the ever-changing COVID-19 variants.
In this climate of uncertainty, vaccination will be key to staying healthy and fighting our way out of the pandemic. “Generally, when the public is well-vaccinated, whether for flu, meningitis, pneumonia, or shingles, we see less disease and less burden on the health care system,” says Dr. Vivien Brown, a family physician in private practice in Toronto and Assistant Professor in the Faculty of Medicine at the University of Toronto.
“With COVID-19 specifically, the vaccines have had a significant impact on driving down the number of hospitalizations and deaths, and in helping to create a cocooning effect to protect people who can’t be vaccinated,” she says.
Ongoing efficacy, adolescent approval, booster shots
Moderna’s efforts are critical in the global fight against COVID-19. To date, almost 146 million doses of Moderna’s COVID vaccine have been administered in the U.S.1 and over 7.2 million have been administered in Canada.2 As the pandemic evolves, Moderna is staying ahead of the science with mRNA technology.
Though the variants, waning immunity, and breakthrough infections are concerning, new data published in Science reported that the majority of individuals vaccinated with the Moderna COVID-19 vaccine maintained antibodies against SARS-CoV-2 variants for six months after the second dose.3
On August 27, 2021, Moderna’s COVID-19 vaccine was authorized by Health Canada for use in adolescent populations aged 12 and above. “We see that as a major win in the fight against COVID-19,” says Dr. Beverly Francis, Director of Scientific Leadership — North America at Moderna. “Vaccinating as many eligible Canadians as possible limits the human pool that serves as a viral reservoir, limits the spread of this virus, and greatly limits the virus’ ability to evolve and mutate into more transmissible or infective strains.”
Anticipating the combination of the force of the current Delta variant, waning immunity, and pandemic fatigue will require further defensive action. Moderna is already preparing by advancing its mRNA-1273 vaccine as a booster candidate.4 “Initial research has found it to be effective in boosting responses broadly against both the ancestral and major variant strains of the virus, and the safety profile of the booster was similar to that observed previously for dose two of the vaccine,” says Dr. Francis.
Finally, to ensure a steady supply, Moderna has been working closely with the Government of Canada to secure supply for up to 105 million doses of the COVID vaccine and its booster candidates, when authorized, for delivery through to 2024.5
mRNA technology as the future of medicine
The Moderna COVID-19 vaccine is the first mRNA vaccine that the company has brought to market. mRNA technology is built on the foundation that our bodies can create their own defence mechanisms. mRNA-based vaccines are designed to deliver the instructions to our cells to create proteins that help activate the immune response against the virus. “It’s like giving the cell a recipe to follow, in this case a recipe to produce a spike protein, which then stimulates the antibodies,” says Dr. Brown.
Moderna’s COVID-19 mRNA vaccines build on more than a decade of basic and applied mRNA science, delivery technology, and manufacturing. The precision, speed, and flexibility of the platform enabled Moderna to respond quickly to the COVID-19 pandemic, providing an effective vaccine in just under a year. “It’s such an elegant and precise platform,” says Dr. Brown. “It’s just so fortuitous that it was ready at a time when we urgently needed it.”
Moderna’s mRNA platform and approach will enable the company to research, develop, and manufacture medicines in new and potentially ground-breaking ways that can help single patients with individualized therapy, or millions of patients with infectious diseases. “We’re essentially industrializing mRNA technology and scaling the scope of therapeutic targets to address diseases, viruses, and pathogens in ways that were previously considered unimaginable,” says Dr. Francis.
Robust R&D investments lead the way to new ways to treat diseases
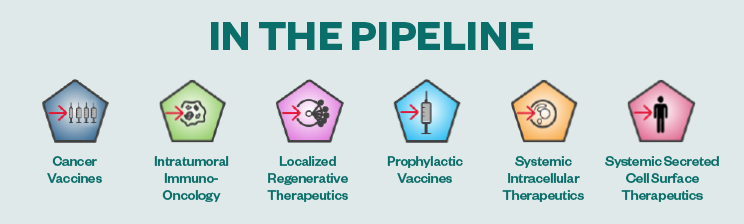
In 2020, Moderna invested $1.37 billion on R&D6 — and it has invested $2.3 billion over the past three years. Moderna’s mRNA pipeline includes 23 projects in development and 15 clinical study programs currently underway.5
Within the infectious disease modality, Moderna currently has nine vaccines in development for major unmet needs, including next-generation vaccines for COVID-19, influenza, cytomegalovirus (CMV), respiratory syncytial virus (RSV), human metapneumovirus (hMPV), parainfluenza (PIV3), and Zika. It has an additional 12 medicines in development across four therapeutic areas — immuno-oncology, rare diseases like propionic acidemia, cardiovascular diseases, and autoimmune diseases.7
“The breadth of the mRNA platform and its capabilities are really energizing,” says Dr. Francis. “If you understand what mRNA is at its core and figure out how to deliver it, as we’ve done, you can see we’re just at the beginning.”
Clinical trials are underway to assess two of Moderna’s new respiratory vaccine candidates — one is a quadrivalent mRNA seasonal flu vaccine candidate, and the other aims to protect against RSV.8 “Our vision is to develop a combination respiratory vaccine for adult and older adult populations, combining seasonal flu, RSV, and COVID-19 booster all in one shot,” says Dr. Francis. “Doing many things in parallel to protect against three serious respiratory viruses at once is typical of the spirit and boldness of Moderna.”
This article was made possible with support from ModernaTx.
1Centres for Disease Control and Prevention. COVID-19 vaccinations in the United States. U.S Department of Health & Human Services: September 2, 2021. https://covid.cdc.gov/covid-data-tracker/#vaccinations
2Public Health Agency of Canada. Canadian COVID-19 vaccination coverage report. Ottawa: Public Health Agency of Canada; August 21, 2021. https://health-infobase.canada.ca/covid-19/vaccination-coverage/
3Moderna press release, August 12, 2021. https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-new-study-showing-its-covid-19-vaccine
4Moderna press release, September 1, 2021. https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-submission-initial-data-us-fda-its-covid-19
5 Moderna press release, August 16, 2021. https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-supply-agreement-canada-expanded-supply
6Moderna press release, February 25, 2021. https://investors.modernatx.com/news-releases/news-release-details/moderna-reports-fourth-quarter-and-fiscal-year-2020-financial
72Q2021 Moderna Earnings Call Business Update presentation, August 5, 2021. https://investors.modernatx.com/static-files/d427592c-dab4-4715-a568-31207d9832ab (slide 9).
82Q2021 Moderna Earnings Call Business Update presentation, August 5, 2021. https://investors.modernatx.com/static-files/d427592c-dab4-4715-a568-31207d9832ab (slide 34).
IMPORTANT SAFETY INFORMATION
SPIKEVAX™ (elasomeran mRNA vaccine) is a vaccine used to prevent the coronavirus disease 2019 (COVID-19) caused by the SARS-CoV-2 virus. It can be given to individuals aged 12 years and older. As with any vaccine, SPIKEVAX may not fully protect all those who receive it. Even after you have had both doses of the vaccine, continue to follow the recommendations of local public health officials to prevent the spread of COVID-19. Individuals may not be optimally protected until after receiving the second dose of the vaccine.
The most common or very common side effects of SPIKEVAX are pain at the injection site, tiredness, headache, muscle ache and stiffness, chills, fever, swelling or redness at the injection site, nausea and/or vomiting and enlarged lymph nodes. Allergic reactions may also occur.
Vaccination may not be suitable for everyone, so ask your healthcare professional if SPIKEVAX is right for you. Full product information can be found on https://www.modernacovid19global.com/ca/. To report an adverse event, please call 1-866-MODERNA (1-866-663-3762).
© 2021 Moderna, Inc.
ISI-0006 09/2021


