
Dr. David Kirsch
Head of the Radiation Medicine Program, The Princess Margaret Cancer Centre

Dr. Jan Seuntjens
Head of Medical Physics at The Princess Margaret Cancer Centre
Thanks to The Princess Margaret, cancer care in Canada continues to evolve and improve.
Cancer care in Canada is among the best in the world. Princess Margaret Cancer Centre is ranked Canada’s top oncology hospital and among the top 10 worldwide.

Mediaplanet spoke with two leaders at The Princess Margaret to learn more about recent developments in radiation medicine. First, we spoke with Dr. David Kirsch, Head of the Radiation Medicine Program, about adaptive radiation therapy and theranostics.
What is adaptive radiation therapy, and why is it a breakthrough for patients?
With adaptive radiation therapy, we get a new scan, either MRI or CT, right before the radiation is delivered, allowing us to modify the radiation plan accordingly. This can limit the radiation dose going to healthy organs while ensuring the tumour is fully targeted.
Which patients are seeing the greatest benefit from adaptive radiation therapy today?
We’re seeing the clearest benefits in cancers where the tumour or nearby organs change position from day to day, such as cancers in the pelvis. Daily imaging allows us to adjust for things like bladder filling or tumour movement, helping protect normal tissues while maintaining full tumour coverage. Adaptive therapy is also being used in other cancers, including head and neck cancers and sarcomas, as we continue to gather data to better understand which patients benefit most.
Theranostics is described as a precision medicine approach. What is it, and how does it expand options for patients?
Theranostics is a way of delivering radiation therapy systemically, usually by intravenous injection, targeting the radiation medicine directly to tumour cells. It can travel throughout the body to treat microscopic disease.
We also spoke with Dr. Jan Seuntjens, medical radiation physicist and Head of Medical Physics at The Princess Margaret, about particle therapy and how AI-enabled workflows are reshaping radiation treatment delivery.
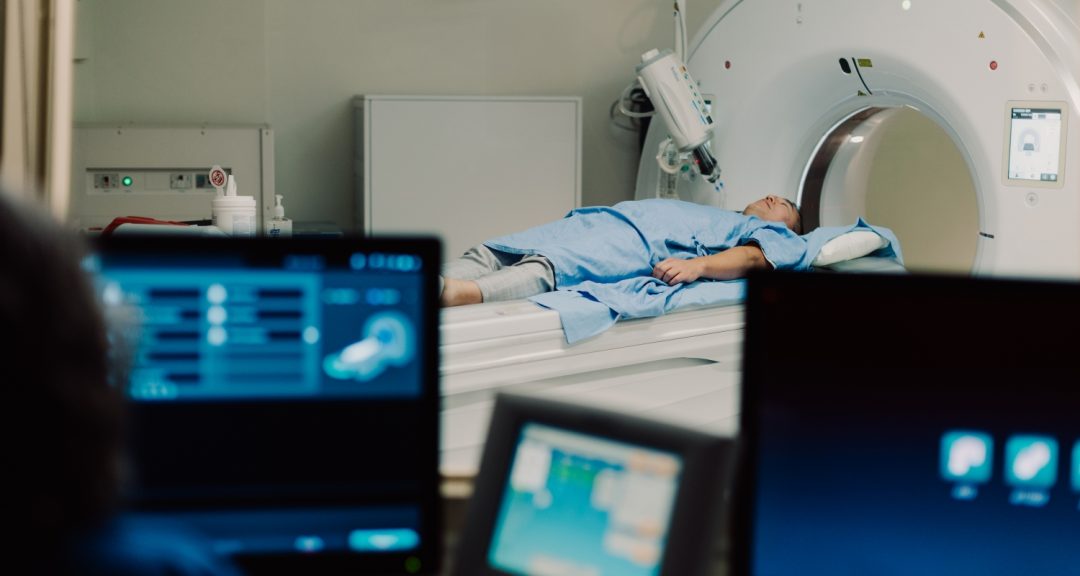
What is particle therapy, and what makes it different from conventional radiation?
With traditional radiotherapy, photon beams deliver the radiation dose to the tumour but also to surrounding tissues as the beam passes through the target. With particle therapy, we can make the particles stop exactly in the tumour target, depositing all oftheir remaining energy, so there’s no radiation dose beyond the target.
How are AI and automation changing treatment planning and day-of delivery?
AI speeds up the segmentation process — outlining the tumour target and the normal tissues we need to spare. AI also automates the radiation planning process, with the plan reviewed and approved by the clinical team before delivery, so treatment can be adapted and adjusted minutes before it’s delivered. What used to take two weeks can now be done within an hour.
With faster AI-enabled workflows, what safeguards and teamwork are needed to deliver treatment safely?
We have three professions who collaborate: the radiation oncologist, the medical physicist, and the radiation therapist. Previously, our workflow followed a strictly sequential model, with tasks passed between professions and quality assurance embedded at specific checkpoints. In the AI-enabled workflow, these quality assurance measures remain fully intact, but the process now emphasizes closer cross-professional collaboration, with shared responsibility for decision-making and verification.
This leadership in cancer care is supported by The Princess Margaret Cancer Foundation, which raises funds to advance research, acquire the most advanced technology, and support patient care, including innovations in radiation medicine such as particle therapy, theranostics, and adaptive therapy.
To learn more, visit thepmcf.ca.




